Recent FDA recalls of Wegovy and Gold Star products underscore ongoing challenges in maintaining sterile manufacturing for injectables and storage. These incidents push the industry toward stricter "Quality by Design" processes to embed safety from the outset, as end-stage defect detection falls short.
Wegovy Recall: Hair Contamination in Prefilled Syringes
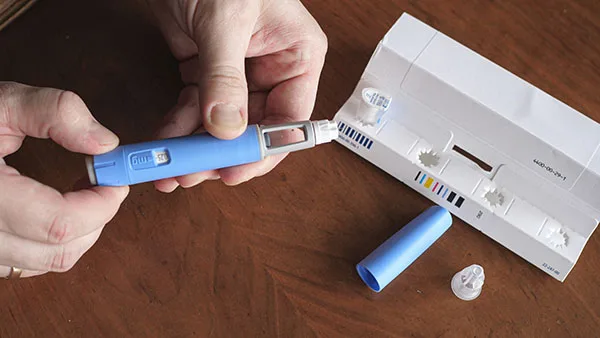
On December 31, the FDA announced recalls for several lots of Wegovy, a GLP-1 prescription injectable for adult weight loss. Inspectors identified "particulate matter"—specifically hair—inside a prefilled syringe.
Novo Nordisk voluntarily recalled specific 0.5 mg and 1 mg single-dose pens. The company stated these were detected during inspection and never distributed to pharmacies or patients. No adverse effects were reported from marketed Wegovy pens, with patient safety as the top priority.
Despite this, hair in an injectable raises alarms. Injectables bypass natural defenses, entering tissue or bloodstream directly. Regulators demand they be free of visible particles.
Risks of Particulate Contamination in Injectables
A strand of hair seems minor outside medicine but signals critical failure in syringes. Known as "particulate contamination," this issue has concerned experts for nearly two centuries, per PharmTech.
Particles can cause:
- Injection site irritation or inflammation
- Immune responses
- Rare blood vessel obstruction
Vulnerable groups—infants, older adults, immunocompromised patients—face elevated risks. The FDA classified this Wegovy recall as Class II, indicating possible temporary or reversible effects with low serious harm probability.
Precision tracking for your journey
Join thousands using Shotlee to accurately track GLP-1 medications and side effects.
📱 Get the Shotlee App
Track your GLP-1 medications, peptides, and health metrics on the go with our mobile app!
Gold Star Distribution Recall: Rodent and Bird Droppings
On December 26, Minneapolis-based Gold Star Distribution recalled all FDA-regulated products from one facility after finding rodent feces, urine, and bird droppings in storage areas.
Affected items included OTC medicines like Advil, Benadryl, DayQuil, NyQuil, and Tylenol, plus cosmetics, supplements, devices, and pet foods. Unsanitary conditions posed risks of microbial contamination, including Salmonella.
No illnesses were reported, but risks include ingestion, skin contact, or handling packaging then touching the face. Salmonella causes abdominal pain, diarrhea, fever, and vomiting; severe cases in vulnerable patients can lead to bloodstream infections and life-threatening complications.
Broader Supply Chain Challenges
These recalls reflect vulnerabilities in pharmaceutical and consumer product chains. PharmTech notes particulate contamination drove about 22% of FDA drug recalls from 2008-2012.
Particulates fall into three categories, each tied to vulnerabilities like environmental controls, equipment wear, human handling, or material interactions:
- Intrinsic (inherent to formulation)
- Extrinsic (from environment/handling)
- Inherent (from interactions)
Complex manufacturing and rising prefilled syringe use increase error opportunities. Proper storage is equally vital—rodents or birds can contaminate surfaces, packaging, and air without breaching seals.
Consumer Lessons from These Recalls
The full chain—from production to storage to shipping—must prioritize safety. Consumers can protect themselves by:
- Checking FDA recall alerts regularly
- Inspecting medications for visible particles before use
- Reporting issues to healthcare providers or FDA
- Storing products in clean, pest-free areas
Staying informed empowers better choices amid complex supply chains for GLP-1 medications like Wegovy.