Hims & Hers Shares Sink 16% as FDA Halts Wegovy Copycat Pill
In a swift regulatory reversal, Hims & Hers Health Inc. (HIMS) has decided to stop selling a copycat version of Novo Nordisk's (NVO) Wegovy weight loss pill, just two days after launching the product. This decision triggered a nearly 16% drop in HIMS shares during after-hours trading. The move stems from mounting pressure from regulators, including a new U.S. Food and Drug Administration (FDA) warning on copycat weight loss drugs, and direct warnings from Novo Nordisk that the copy was illegal.
The Immediate Fallout: Hims Pulls the Plug on Wegovy Copycat
Hims & Hers announced the halt following "constructive conversations with stakeholders," as stated in a post on X. The company faced dual pressures: Novo Nordisk threatened legal action, and the Department of Health and Human Services referred Hims to the Department of Justice for potential federal law violations. Neither Novo nor the FDA provided immediate additional comments.
This incident highlights the precarious position of telehealth companies venturing into compounded versions of blockbuster GLP-1 receptor agonists like Wegovy (semaglutide). Wegovy, approved for chronic weight management, has driven massive demand in the obesity treatment market, but supply constraints previously opened doors for alternatives.
Background on Compounded GLP-1 Drugs and Wegovy
GLP-1 medications, such as Novo Nordisk's Wegovy and Ozempic (also semaglutide) or Eli Lilly's (LLY) Zepbound (tirzepatide), mimic hormones that regulate blood sugar and appetite. They promote significant weight loss—often 15-20% of body weight in clinical trials—making them a cornerstone of metabolic health therapy.
For years, telehealth firms like Hims & Hers have relied on compounding pharmacies to produce lower-cost versions of these drugs during FDA-declared shortages. Compounding involves customizing medications, which was permissible under Section 503A of the Federal Food, Drug, and Cosmetic Act when branded supplies were insufficient. This practice exploded as patient demand surged post-2021, with compounded semaglutide filling gaps.
However, as shortages have eased—Novo ramped up production, resolving the Wegovy shortage by late 2023—regulators have signaled a crackdown. The FDA's recent warning explicitly targets unauthorized compounded weight loss drugs, emphasizing safety risks like inconsistent dosing and contamination not present in FDA-approved products.
How Compounded Wegovy Copycats Differ from the Brand
- Branded Wegovy: FDA-approved, with precise dosing (starting at 0.25 mg weekly, titrating to 2.4 mg), rigorous quality control, and established cardiovascular safety data.
- Compounded Versions: Often cheaper ($200-400/month vs. $1,300+ for branded), but lack standardization, potentially leading to variable efficacy or adverse effects like nausea, gastrointestinal issues, or rare pancreatitis.
Patients using compounded GLP-1s should monitor for side effects closely; apps like Shotlee can help track symptoms, dosages, and progress during therapy.
Regulatory Shift: FDA and Big Pharma Push Back
Novo Nordisk and Eli Lilly have long argued the FDA hasn't done enough to curb knockoffs, which they claim undermine innovation and patient safety. This Hims case may signal a turning point: it's the first instance of a telehealth firm pulling a copycat product after a direct company complaint.
The FDA's stance aligns with broader efforts to enforce drug approval pathways. Post-shortage, compounding for "essentially a copy of a commercially available drug" violates regulations, per FDA guidance. The HHS-DOJ referral underscores potential criminal implications for non-compliance.
Precision tracking for your journey
Join thousands using Shotlee to accurately track GLP-1 medications and side effects.
📱 Get the Shotlee App
Track your GLP-1 medications, peptides, and health metrics on the go with our mobile app!
Past Tensions Between Hims and Novo
Hims & Hers has history with Novo. Last year, they explored a partnership for discounted weight loss shots, but it collapsed. Novo executive Ludovic Helfgott noted, "We had an agreement that the mass compounding would stop, and unfortunately, it didn't stop." Hims CEO Andrew Dudum had vowed not to "cave" to pharma demands, yet the company retreated here.
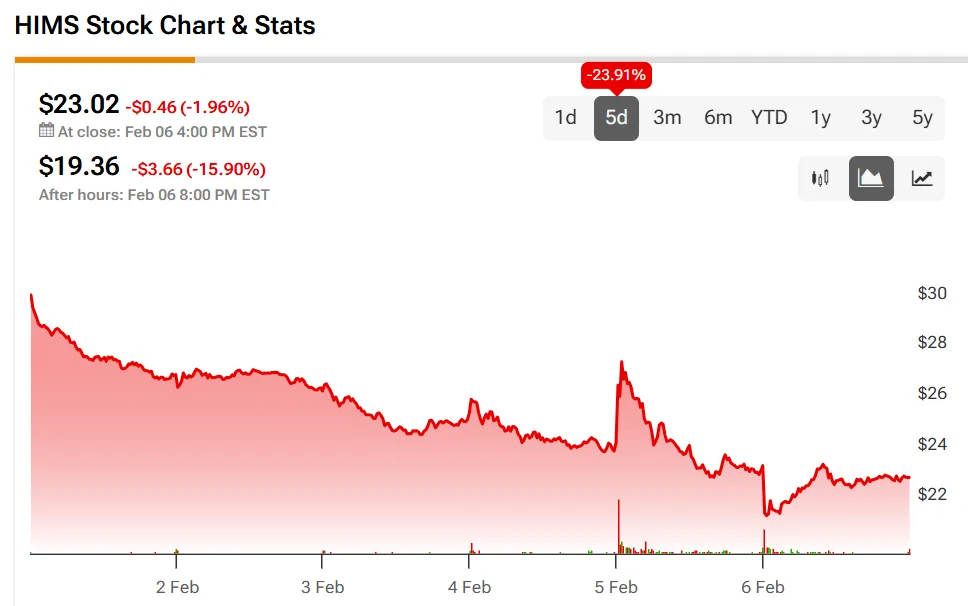
Stock Market Impact on Hims & Hers (HIMS)
The announcement hit HIMS shares hard, down nearly 16% in after-hours trading at the time of writing. Hims has aggressively expanded into the weight loss market via telehealth, offering virtual consultations and prescriptions. This setback underscores challenges in competing against pharma giants and navigating regulations.
Analysts maintain a Hold consensus on HIMS, with an average price target of $38.14—implying 65.68% upside from recent levels. Investors eye telehealth's growth but factor in regulatory risks.
Broader Implications for the Weight Loss Market and Telehealth
The GLP-1 market, projected to exceed $100 billion by 2030, faces transformation. With shortages resolved, branded drugs regain dominance, potentially limiting affordable access. Telehealth providers must pivot: some shift to FDA-approved generics or lifestyle programs, while others explore novel peptides.
For patients, this means discussing options with providers. Branded GLP-1s offer insurance coverage for eligible obesity or diabetes cases, unlike many compounded alternatives. Alternatives include oral semaglutide (Rybelsus), tirzepatide (Zepbound/Mounjaro), or non-drug metabolic therapies.
Safety Considerations for GLP-1 Users
- Common Side Effects: Nausea, vomiting, diarrhea—often transient with slow titration.
- Serious Risks: Thyroid tumors (boxed warning), gastroparesis; compounded versions amplify uncertainties.
- Monitoring Tips: Regular bloodwork, hydration, and tools for symptom logging.
What This Means for Patients Seeking Weight Loss Treatments
If you're exploring GLP-1 therapy, prioritize FDA-approved options like Wegovy. Consult your doctor about eligibility (BMI ≥30 or ≥27 with comorbidities), titration schedules, and cost-saving programs (e.g., Novo savings cards). Telehealth remains valuable for access but verify prescription legitimacy.
This Hims episode reinforces: compounded drugs filled a critical gap but aren't substitutes long-term. As regulations tighten, innovation in peptide therapy and metabolic health may accelerate safer, accessible alternatives.
Key Takeaways
- Hims & Hers halted Wegovy copycat sales two days post-launch due to FDA crackdown, Novo threats, and DOJ referral.
- Shares dropped 16%; analysts Hold with $38.14 target.
- Compounding boomed during shortages but faces limits now.
- Patients: Opt for approved GLP-1s, monitor closely, discuss with providers.
- Market shift favors big pharma, challenges telehealth expansion.
In summary, this regulatory action protects patients from unverified compounds while reshaping competition in GLP-1 weight loss drugs. Stay informed on updates from FDA and manufacturers for the latest on safe metabolic health options.