Semaglutide: Artificial Shortage as Novo Nordisk's Business Model?
In early February, drug company Novo Nordisk sued telehealth company Hims & Hers, spotlighting tensions in the semaglutide market. The core issue revolves around compounded versions of semaglutide—the active ingredient in Novo Nordisk's Wegovy—being sold at significantly lower prices. This legal battle raises questions about access to GLP-1 medications, patent protections, and whether artificial shortages are being engineered to sustain premium pricing.
The Novo Nordisk vs. Hims & Hers Lawsuit: What Happened?
The complaint from Novo Nordisk accuses Hims & Hers of selling GLP-1 drugs like semaglutide at lower prices than the branded versions. At its simplest, it's a clash over market share. But the legal details hinge on the U.S. Food and Drug Administration's (FDA) "Drug Shortage List." Semaglutide, the drug behind Novo Nordisk's Wegovy brand, was removed from this list more than a year ago.
While on the shortage list, compounding pharmacies like those partnered with Hims & Hers could produce semaglutide under a special legal dispensation, bypassing Novo Nordisk's patent. Post-removal, Novo Nordisk argues these pharmacies no longer have permission to produce it without infringing on their "Very Special Important Government-Granted Monopoly, aka a patent," on the substance.
"This is a complete sham, and it has been a sham since the shortage ended," Novo Nordisk attorney John Kuckelman tells CNBC. "The fact is that their medicines are untested, and they're putting patients at risk."
This quote from Kuckelman highlights safety concerns, though the chemical compound—semaglutide—remains identical between branded and compounded versions; the primary difference is the label and branding.
Understanding Compounding Pharmacies and GLP-1 Access
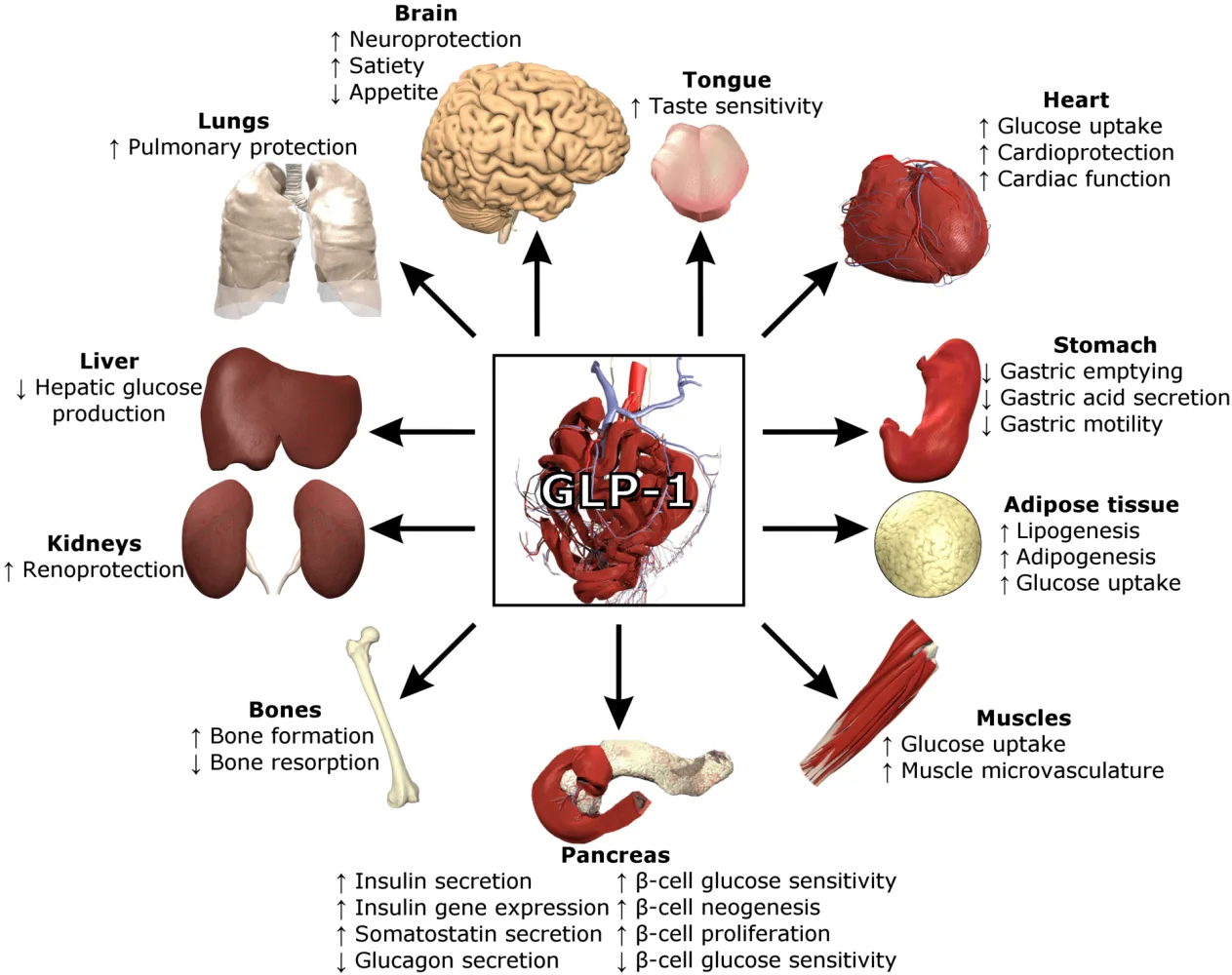
Compounding pharmacies customize medications to meet specific patient needs, often during shortages. For GLP-1 agonists like semaglutide, used primarily for type 2 diabetes management and chronic weight loss, this has been crucial. Semaglutide mimics glucagon-like peptide-1 (GLP-1), a hormone that regulates blood sugar, slows gastric emptying, and promotes satiety, leading to weight loss and improved metabolic health.
Patients turning to compounded options often do so for affordability or availability, especially as demand surges. However, post-shortage enforcement aims to redirect patients to branded products like Wegovy or Ozempic injections.
Semaglutide Pricing: Branded Wegovy vs. Compounded Alternatives
Novo Nordisk recently introduced Wegovy in pill form, priced at about $150 a month. In contrast, Hims & Hers planned to offer the same chemical compound—semaglutide—without Wegovy branding for around $50 a month. This stark difference underscores the lawsuit's stakes.
The enforcement of Novo Nordisk's patent on semaglutide in pill form is designed to create a shortage of affordable options. By limiting competition, the company can charge at least three times the market-driven profitable price. Hims & Hers' $50 offering demonstrates that production costs allow for lower pricing in a competitive environment.
Why Prices Differ: The Economics of Perfect Competition
In a theoretical "perfect competition" model, prices align closely with production costs because sellers must offer the lowest price to capture market share. Real-world factors like shipping, marketing, and branding influence prices, but most consumers wouldn't pay three times more for identical semaglutide from Novo Nordisk versus Hims & Hers.
Precision tracking for your journey
Join thousands using Shotlee to accurately track GLP-1 medications and side effects.
📱 Get the Shotlee App
Track your GLP-1 medications, peptides, and health metrics on the go with our mobile app!
For patients with obesity or diabetes, this pricing gap affects treatment adherence. High costs lead to rationing doses or discontinuation, undermining GLP-1 benefits like sustained weight loss (up to 15-20% body weight in trials) and cardiovascular risk reduction.
The Role of Patents in Semaglutide and GLP-1 Markets
Patents grant inventors exclusive rights for a limited time, as per the U.S. Constitution, to "promote the progress of science and useful arts." In practice, this creates artificial shortages by preventing competitors from copying inventions.
Novo Nordisk has benefited from being first to market with semaglutide, succeeding against other GLP-1 products from competitors like Eli Lilly's tirzepatide (Mounjaro/Zepbound). Yet, the company seeks government intervention to eliminate lower-cost rivals entirely. This strategy prioritizes monopoly profits over broader access.
Patents vs. Patient Access: A GLP-1 Test Case
Amid hype around GLP-1s for weight loss, diabetes, heart health, and even mental conditions, semaglutide exemplifies patent tensions. While benefits are real—improved glycemic control, reduced A1C, and appetite suppression—skepticism exists for the most extravagant claims. Regardless, granting monopolies enables price gouging on proven therapies.
Patients should weigh branded reliability against compounded affordability, consulting providers about FDA-approved options versus custom formulations.
Safety Considerations for Semaglutide Users
Novo Nordisk's attorney raised alarms about untested compounded medicines risking patients. Branded Wegovy undergoes rigorous FDA trials for purity, dosing accuracy, and stability. Compounded versions, while using the same semaglutide, may vary in quality control.
Common GLP-1 side effects include nausea, vomiting, diarrhea, and potential thyroid risks. Patients monitoring symptoms—perhaps with apps like Shotlee for tracking side effects and schedules—can better manage therapy. Always discuss compounding with a healthcare provider to ensure safety.
What This Means for Patients Seeking GLP-1 Therapy
Who Might Benefit from Semaglutide?
Ideal candidates include adults with BMI ≥30 (obesity) or ≥27 with comorbidities like hypertension or prediabetes. Semaglutide's once-weekly injection or new oral form offers convenience over daily pills.
Practical Guidance: Discussing Options with Your Doctor
Ask about branded vs. compounded semaglutide availability, insurance coverage (often better for Wegovy/Ozempic), and cost-saving programs. Explore alternatives like liraglutide or tirzepatide if semaglutide shortages persist.
- Verify pharmacy compounding standards (PCAB accreditation).
- Monitor for shortages via FDA updates.
- Combine with lifestyle changes for optimal results.
Key Takeaways
- Novo Nordisk sued Hims & Hers in February over post-shortage compounded semaglutide sales.
- Wegovy pills cost $150/month vs. $50 compounded alternatives, highlighting patent-driven pricing.
- Patents create artificial monopolies, limiting competition despite market viability at lower prices.
- Safety: Branded products offer tested assurance; compounded require caution.
- Patients: Prioritize doctor consultations for affordable, safe GLP-1 access.
Conclusion: Balancing Innovation, Access, and Affordability
The Novo Nordisk-Hims & Hers dispute reveals how patent enforcement on semaglutide perpetuates artificial shortages as a business model. While protecting innovation, it inflates costs for life-changing GLP-1 therapies. Patients deserve transparent options—branded reliability or compounded savings—without monopolistic barriers. Stay informed on FDA shortage status and advocate for policies promoting competition in metabolic health treatments.