The Problem with Search Results
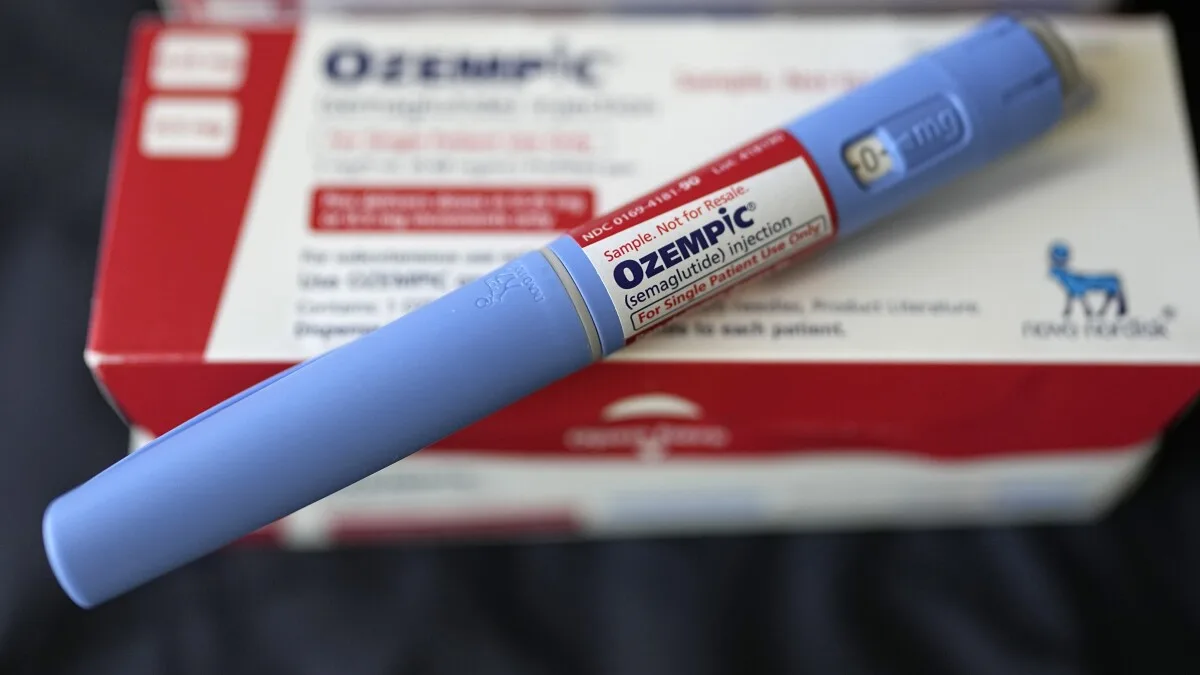
If you've entered 'weight loss' into a search engine, chances are high that a site for Ozempic popped up among the initial listings.
Yet, Ozempic does not have FDA approval for weight loss; its sanctioned use is solely for managing Type 2 diabetes. What leads to this visibility?
The explanation lies in sponsored search listings. Businesses compensate search platforms to elevate their pages as top results for specific terms or expressions.
'Search engines frequently serve as the initial stop for individuals seeking answers to health inquiries,' notes Daniel Eisenkraft Klein, a research fellow affiliated with Harvard Medical School and Brigham and Women's Hospital's Program on Regulation, Therapeutics and Law.
'Pharma firms have mastered manipulating this via pay-per-click promotions, effectively purchasing prime positions in search listings,' he continued.
Advertising Regulations Compared
Typically, pharmaceutical entities must adhere to rigorous guidelines for promoting products in print media or on TV.
These include revealing potential risks or adverse reactions, and prohibiting promotion for unapproved treatments, even if off-label prescribing occurs in practice.
Violations can result in FDA penalties, aimed at avoiding deceptive content that might endanger people.
However, online sponsored listings, often positioned above organic results, lack the same regulatory oversight. Legislation has not evolved to match technological shifts.
Study Insights on Ozempic
Eisenkraft Klein and colleagues examined two years of paid listings for Ozempic, funded by manufacturer Novo Nordisk.
Their findings appeared in JAMA Network Open.
Analysis revealed that 11 percent of paid keywords included 'weight,' despite Ozempic's absence of weight loss approval.
Additionally, Novo Nordisk invested in listings for terms referencing rivals like Trulicity and Mounjaro from Eli Lilly.
Precision tracking for your journey
Join thousands using Shotlee to accurately track GLP-1 medications and side effects.
📱 Get the Shotlee App
Track your GLP-1 medications, peptides, and health metrics on the go with our mobile app!
'Novo Nordisk funded the query 'Kelly Clarkson weight loss,' likely targeting those interested in celebrity weight management solutions,' explained Eisenkraft Klein.
The team concentrated solely on Ozempic, and further investigation into sponsored listings for other medications remains necessary.
Consumer Advice
Nevertheless, Eisenkraft Klein views the research as instructive for users: 'Do not presume Google presents the most pertinent data. Top placement does not guarantee objectivity or relevance.'
He advises bypassing sponsored listings and consulting unbiased outlets, including university medical institutions, for online drug research.
Novo Nordisk representative Liz Skrbkova stated via email that the study inaccurately depicts the firm's 'paid search strategy,' which employs 'standard industry' methods to connect with users.
The company asserts adherence to U.S. laws, regulations, and internal ethics to enable patient access to medication details for collaborative healthcare choices.
Broader Context
The U.S. stands out as one of few nations permitting direct consumer promotion of prescription medications.
In September, the Trump administration issued a directive urging FDA action on lax risk disclosures in drug promotions.
The agency also issued multiple warning letters to firms regarding their advertisements.
An FDA commitment ensures promotional materials for prescription drugs remain 'truthful, balanced, and accurately conveyed,' per a statement from Department of Health and Human Services spokesperson Emily Hilliard. She recommends reporting issues to the agency's Bad Ad Program.
Google spokesperson Nate Funkhouser declared in a statement that the company maintains strict advertising guidelines for prescription drugs: 'Advertisers bear full responsibility for aligning their strategies, including keywords and content, with legal mandates.'
Health tracking apps like Shotlee can assist in monitoring weight loss progress alongside informed decisions.