Peptides are gaining recognition for their potential in optimizing health and wellness. Recent research sheds light on how specific leader peptides interact with enzymes, revealing possibilities for developing novel therapies and improving overall well-being. Tools like Shotlee can help you track your peptide therapy progress (GLP-1, BPC-157, melanotan, thymosin, etc.) alongside other crucial health metrics.
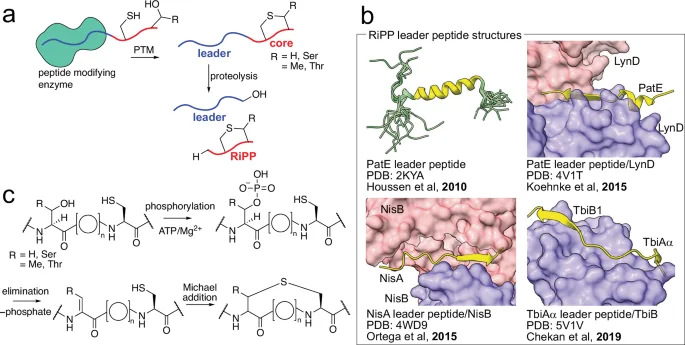
A groundbreaking study published in *Nature Communications* explored leader peptides and their interaction with RiPP halogenases and lanthionine synthetases. The research focused on LanM enzymes, which modify precursor peptides containing a specific structure called an NHLP. Scientists aimed to identify new LanM substrates to understand the natural products they create and their specific functions. They also sought to pinpoint common characteristics across diverse NHLP-modifying LanMs. The study identified a Desulfotomaculum sp.-derived proteusin RiPP BGC (dpr BGC) containing seven NHLP-containing precursor peptides. These DprE peptides possess conserved leader sequences but variable core sequences, suggesting diverse potential functions.
The researchers successfully co-expressed DprM, a lanthionine synthetase, with DprE peptides, leading to the formation of (methyl)lanthionine rings. This demonstrated DprM's tolerance for different core sequences and its ability to generate rings of varying sizes. Furthermore, DprM exhibited selectivity for specific leader peptides, highlighting the importance of these sequences in enzyme recognition and binding. The team also found that the core peptide has minimal effect on the NHLP structure.
Precision tracking for your journey
Join thousands using Shotlee to accurately track GLP-1 medications and side effects.
📱 Get the Shotlee App
Track your GLP-1 medications, peptides, and health metrics on the go with our mobile app!
What does this mean for your health journey? Understanding these peptide interactions opens doors to designing new peptides with targeted effects. For example, BPC-157 is a peptide known for its potential healing properties, while GLP-1 receptor agonists like semaglutide are used for weight management. With Shotlee, you can monitor the impact of such peptide therapies on your weight loss goals, sleep patterns, and other health metrics. This data-driven approach empowers you to fine-tune your wellness strategy for optimal results. The ability to track subtle changes in your health provides valuable insights that can improve your longevity and overall quality of life.
By leveraging health tracking tools like Shotlee, individuals can proactively manage their wellness journey, making informed decisions about peptide therapies and other interventions based on personalized data. Whether you're exploring the potential of melanotan, thymosin, or other peptides, Shotlee allows you to monitor your progress, track side effects, and achieve your health and wellness objectives more effectively.