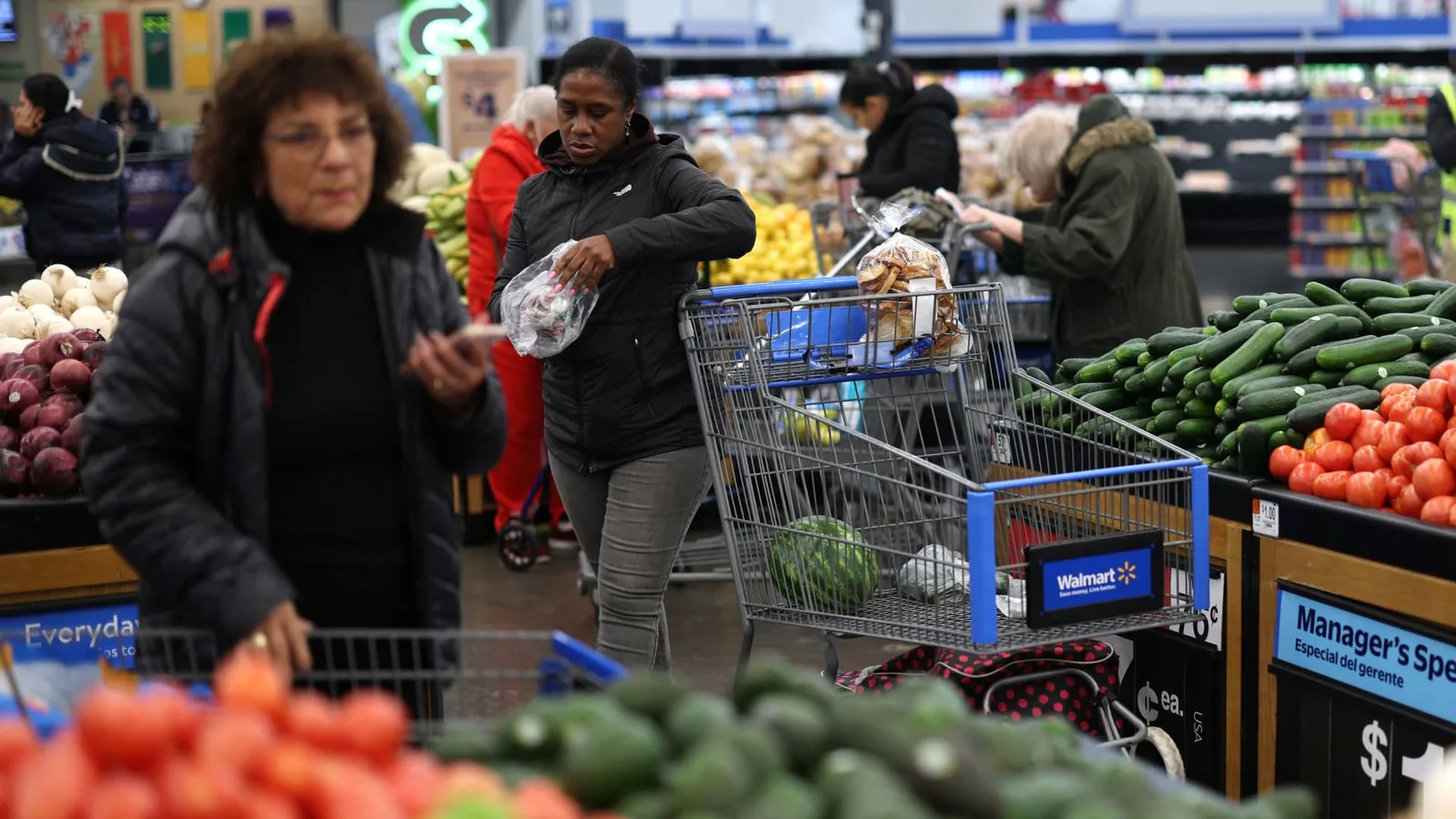The Shift Towards Widespread Adoption
A minor disturbance is poised to transform into a significant surge. Currently, multiple obstacles hinder individuals from addressing obesity through GLP-1 medications. Initially, production challenges led to insufficient availability of Novo Nordisk's Wegovy and Eli Lilly's Zepbound. Coverage problems with insurers persisted for these costly therapies, especially among seniors on Medicare, which prohibits funding for weight loss remedies. Additionally, apprehension existed: Many individuals dislike injections and reject weekly shots. As 2026 unfolds, numerous barriers are eroding. Costs are decreasing, coverage is expanding, and pill forms are anticipated to launch. This indicates a rise in users of GLP-1 treatments. It's positive for overall well-being. Yet, it will profoundly alter consumer routines, opening doors for financial gains.
Expanding User Base
Almost one-fourth of United States households have utilized GLP-1 medications either temporarily or periodically, per research from Circana released in November. The organization noted that most individuals use these drugs for half a year to a year, with just 48.2% continuing treatment fully. Expense ranked as the top factor for stopping, according to the study. Since weight often returns upon cessation, usage periods might extend as expenses drop and oral versions, which are cheaper and simpler, become available.
"Drawing from our latest data, we foresee 35% of food and beverage items and 37% of nonfood items originating from GLP-1-using homes by 2030, assuming current patterns persist," stated Circana in their analysis. Bernstein pharmaceuticals expert Courtney Breen anticipates distinct market niches developing with additional GLP-1 offerings. These niches will mirror user situations, such as employing incretin therapies for extreme obesity, cosmetic purposes, or other ailments. "At present, GLP-1 receptor activators are sanctioned for type 2 diabetes and obesity control (plus related conditions), but their path likely continues," Breen remarked in a December 11 research update. "The GLP-1 receptor appears extensively beyond the pancreas, notably in lungs, heart, brain, kidneys, and digestive system. Based on this science and existing proof, leading firms Eli Lilly and Novo Nordisk (not under coverage) explore further uses for GLP-1 in various medical fields."
In another analysis, Breen highlighted Lilly's oral GLP-1, orforglipron, as an excellent choice for those reducing from injectable options, potentially simplifying long-term weight control. These views came after Lilly disclosed clinical results mid-December. The Food and Drug Administration evaluates Lilly's data, with experts predicting approval around March or April. Novo Nordisk's oral GLP-1 gained FDA endorsement late December, slated for release early January. Lilly's stock concludes 2025 with roughly 40% growth, becoming the inaugural $1 trillion healthcare share. Novo Nordisk, despite dominance, faced weak earnings and declined nearly 40%.
Precision tracking for your journey
Join thousands using Shotlee to accurately track GLP-1 medications and side effects.
📱 Get the Shotlee App
Track your GLP-1 medications, peptides, and health metrics on the go with our mobile app!
Emerging Lifestyle Changes
GLP-1 medications foster novel routines. Oprah Winfrey, who started these drugs over two and a half years ago, shared her journey in a People interview published Tuesday. The media titan and ex-host promotes a co-authored book on obesity with Dr. Ania Jastreboff, detailing her renewed positivity and behaviors, including abstaining from alcohol. Winfrey also mentioned the disappearance of constant food cravings. Circana's data supports this. The researcher observed changes in shopping selections—fewer groceries and drinks, but increased purchases of oral hygiene and personal care items. GLP-1 users continue eating out, yet they favor protein-heavy dishes, controlled portions, and fiber-rich meals, as per their findings.
"This presents chances for eatery models that adapt menus to include healthier, protein-focused, and reduced-size selections," noted Goldman Sachs restaurant analyst Christine Cho in her 2026 forecast. She referenced Cheesecake Factory's bowls and bites, Darden Restaurant's portion experiments, and protein-enriched brews from Dutch Bros and Starbucks as adaptive strategies. According to Cho, a typical GLP-1 consumer allocates more funds to informal dining spots perceived as nutrient-dense. Bonnie Herzog, a Goldman consumer staples analyst, identifies a similar pattern for packaged food and drink firms, risking those lacking healthy alternatives. In her client outlook for 2026, Herzog highlighted NielsenIQ statistics showing accelerated sales for items near store edges, where fresh produce, meats, and seafood reside.
"We consider this linked to heightened emphasis on nutritious eating, and growing GLP-1 uptake might further enlarge the divide as buyers move from processed foods to fresh or chilled options," Herzog explained. She also predicted reduced alcohol intake. This trend benefits Albertsons, Kroger, and Sprouts Farmers Market, along with protein producers Hormel Foods, Smithfield Foods, and Tyson Foods. Consumer goods firms are adjusting, and time will reveal success. Herzog mentioned PepsiCo's innovations in functional drinks, like Muscle Milk sans artificial additives, Starbucks Coffee & Protein, and Propel Protein Water. Pepsi shares end 2025 with over 5% decline, yet Herzog deems the pessimism "excessive." Dining and consumer goods equities lagged the market in 2025. The coming year won't offer simple recovery for these sectors, per Goldman experts. Still, they see potential for certain shares to excel. Cho views GLP-1-induced routines as an "evolving" idea for eateries, with performance more influenced by broader factors like cost-effectiveness and expansion ability.
"We advise placing fresh capital in equities tied to high-growth, lucrative areas that surpass general Staples, such as energy beverages, nicotine products, confections, and cosmetics," Herzog recommended. Health tracking apps like Shotlee can help monitor these new patterns.