Benefits of GLP-1 in Type 1 Diabetes
Utilizing GLP-1 receptor agonists or other incretin-based medications correlated with reduced chances of hospital stays and emergency department visits among adults suffering from type 1 diabetes.
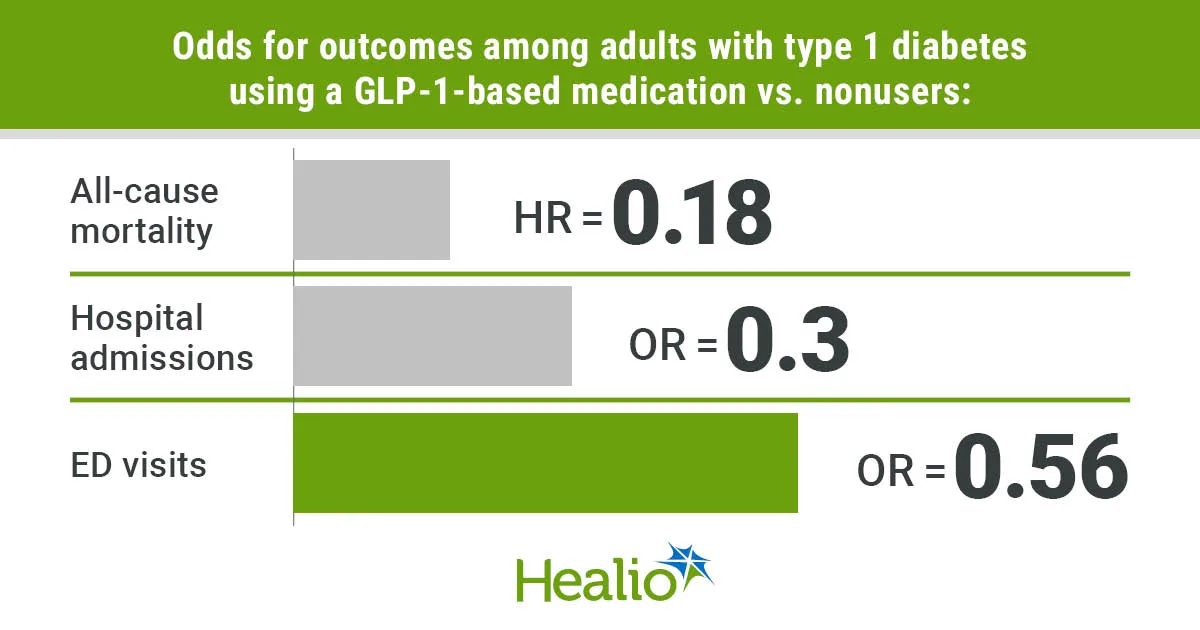
Individuals diagnosed with type 1 diabetes who took a GLP-1 receptor agonist or similar incretin drug experienced a decreased likelihood of overall death, admissions to hospitals, and various other health-related results compared to those who did not use such a medication, as per research findings.
Samita Garg, MD, an associate professor in medicine at Cleveland Clinic Lerner College affiliated with Case Western Reserve University, commented on the implications.
Emerging Advantages for Type 1 Patients
Recent investigations propose that incretin drugs, originally created for type 2 diabetes, could provide substantial advantages to adults living with type 1 diabetes. The findings, shared during the American Diabetes Association's 84th Scientific Sessions, point to diminished chances of death and hospital stays in type 1 diabetes patients receiving these treatments.
Scientists examined information from a sizable group of people with type 1 diabetes, opposing those who received an incretin drug—including semaglutide (Ozempic/Wegovy from Novo Nordisk), tirzepatide (Mounjaro/Zepbound from Eli Lilly), liraglutide (Victoza/Saxenda from Novo Nordisk), dulaglutide (Trulicity from Eli Lilly), albiglutide (Tanzeum from GlaxoSmithKline, no longer available in the U.S.), exenatide (Byetta/Bydureon from AstraZeneca, discontinued in the U.S.), and lixisenatide (Adlyxin from Sanofi-Aventis, discontinued in the U.S.)—against a control group that had not tried such a medication.
Precision tracking for your journey
Join thousands using Shotlee to accurately track GLP-1 medications and side effects.
📱 Get the Shotlee App
Track your GLP-1 medications, peptides, and health metrics on the go with our mobile app!
After accounting for possible interfering variables through propensity score matching, the team discovered that individuals on an incretin drug showed markedly reduced risks of all-cause death (HR = 0.18; 95% CI, 0.11-0.3; P < .0001), lower probabilities of hospital entry (OR = 0.3; 95% CI, 0.2-0.45; P < .0001), emergency department visits (OR = 0.56; 95% CI, 0.43-0.71; P < .0001), endoscopic procedures (OR = 0.52; 95% CI, 0.38-0.7; P < .0001), prescriptions for prokinetics (OR = 0.74; 95% CI, 0.57-0.96; P = .0238), and prescriptions for laxatives (OR = 0.52; 95% CI, 0.43-0.63; P < .0001).
Consistency Across BMI Levels
The advantages held steady throughout various BMI ranges. Non-obese adults, as well as those with class I obesity (BMI: 30-34.99 kg/m²) and class II obesity (BMI: 35-39.99 kg/m²), all saw fewer hospital stays, emergency visits, endoscopic interventions, prokinetic prescriptions, and laxative prescriptions while on an incretin drug.
Moreover, individuals with any degree of obesity faced lower chances of requiring antiemetics when prescribed an incretin medication. In those with class III obesity (BMI: 40 kg/m²), the treatments linked to reduced endoscopic procedures and laxative prescriptions.
Experts stress the importance of additional studies. "Extended observational research will prove crucial" to evaluate the long-term safety and effectiveness of these medications in the type 1 diabetes community, stated Dr. Garg.
As off-label applications of these substances increase, especially among those with obesity or insulin resistance, more research remains necessary. Health tracking apps like Shotlee can assist in monitoring these outcomes for diabetes management.