Understanding the Link Between Sleep Deprivation and GLP-1 Response
Acute sleep deprivation has emerged as a critical factor influencing metabolic health, particularly through its effects on glucagon-like peptide 1 (GLP-1), a key incretin hormone. A study published in Nutrition & Diabetes titled "Acute sleep deprivation delays the glucagon-like peptide 1 peak response to breakfast in healthy men" demonstrates how total sleep deprivation (TSD) disrupts the normal post-meal GLP-1 surge. This research is especially relevant for those interested in GLP-1 medications, peptide therapy, and strategies to optimize metabolic function, as it sheds light on endogenous GLP-1 dynamics under sleep stress.
GLP-1 plays a vital role in enhancing insulin secretion after nutrient intake, suppressing glucagon, slowing gastric emptying, and reducing food intake. Disruptions in its secretion, such as those induced by sleep loss, could contribute to the increased risks of obesity and type 2 diabetes observed in epidemiological data. Prior studies have shown acute sleep loss boosts food intake and impairs glucose tolerance and insulin sensitivity, prompting investigators to explore GLP-1's involvement.
Background: Sleep Loss and Metabolic Disruptions
Epidemiological observations consistently link short sleep durations to heightened risks of obesity and type 2 diabetes. Experimental evidence supports this: acute sleep deprivation elevates food intake and compromises glucose tolerance and insulin sensitivity. As an incretin, GLP-1 is secreted postprandially to amplify insulin release and curb appetite in humans.
Previous research offers mixed insights. One study found reduced afternoon plasma GLP-1 after fragmented sleep in healthy males, though 24-hour levels remained unchanged—possibly due to the mild intervention shifting REM to stage 2 sleep. Another reported decreased GLP-1 in women (but not men) after three nights of 4-hour sleep restriction. These inconsistencies underscore the need for controlled examinations of acute sleep deprivation's impact on GLP-1 profiles, setting the stage for the current investigation into 24-hour serum GLP-1 in healthy young men under regular sleep versus TSD.
Why This Matters for GLP-1 Signaling
GLP-1's dual role in glycemic control and satiety makes it a cornerstone of metabolic health. In clinical practice, GLP-1 receptor agonists like semaglutide (Ozempic, Wegovy) mimic these effects for diabetes and weight management. Understanding how sleep deprivation blunts natural GLP-1 responses could inform patient counseling on lifestyle factors that enhance therapy efficacy, such as prioritizing sleep hygiene alongside peptide therapy.
Study Design and Methodology
The study involved twelve healthy men (mean ± s.e.m., age = 21.9 ± 0.7 years; BMI = 24.1 ± 0.6 kg/m²) with regular sleep-wake rhythms over the prior 6 weeks and no medications. Acute illness was ruled out via physical exams and lab tests. Participants provided written informed consent, adhering to the Declaration of Helsinki and local ethics approval. Data came from a larger study on sleep deprivation's energy expenditure effects, excluding two subjects due to technical issues.
Each participant completed two 24-hour lab sessions—sleep and TSD—separated by 4 weeks, following an adaptation night. From 1800 hours, subjects rested supine in bed until 1300 hours the next day, then sat. In the sleep condition, polysomnography (Nihon Kohden GmbH) monitored sleep from 2300 to 0700 hours, scored per standard criteria. TSD kept subjects awake under ~300 lux light, allowing non-arousing activities like movies, games, or reading under supervision.
Physical activity and intake were standardized. Meals included: 1930 hours (~1.7 MJ; carbs 0.7 MJ, fat 0.5 MJ, protein 0.5 MJ); 0830 hours breakfast (~3.8 MJ; carbs 1.9 MJ, fat 1.3 MJ, protein 0.6 MJ)—specifically 600 ml Fresubin energy drink (112.8 g carbs) at 20 ml/min; and 1330 hours (~4.5 MJ; carbs 1.9 MJ, fat 1.9 MJ, protein 0.7 MJ). A 16.5 MJ ad libitum buffet followed at session end. Water was unlimited; no extra food allowed.
Blood was sampled every 1.5-3 hours in EDTA tubes, including via long thin tubes during sleep to avoid disturbance. Samples centrifuged (2000 g, 10 min) and stored at -80°C. Total GLP-1 measured via Millipore radioimmunoassay (lowest detectable 3 pmol/l with 300 μl extract; inter-assay CV 23%, intra-assay CV 22%). Data normalized to 24-hour individual means; analyzed with repeated-measures ANOVA (Sleep/TSD and Time factors), post-hoc t-tests/Wilcoxon, and trapezoidal AUC. Significance at P < 0.05.
Precision tracking for your journey
Join thousands using Shotlee to accurately track GLP-1 medications and side effects.
📱 Get the Shotlee App
Track your GLP-1 medications, peptides, and health metrics on the go with our mobile app!
Controlled Conditions Ensure Robust Findings
This rigorous setup isolates sleep's impact, controlling for confounders like activity, light, and nutrition—crucial for replicability in metabolic research.
Key Findings: Delayed GLP-1 Peak After Breakfast
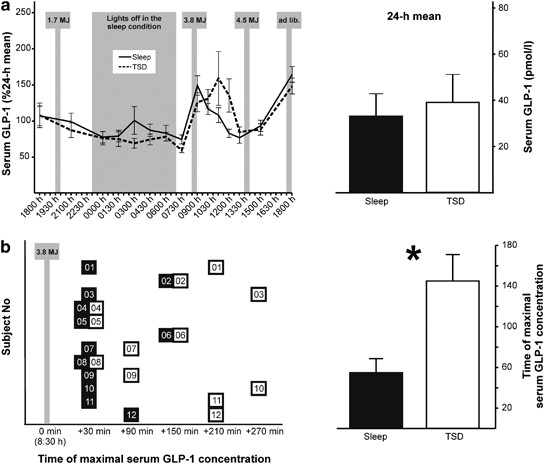
The study's core revelation aligns with its title: acute sleep deprivation delays the glucagon-like peptide 1 peak response to breakfast in healthy men. Under TSD, the post-breakfast GLP-1 surge—normally prompt after the 0830 hours carb-rich meal—was postponed compared to regular sleep. While full 24-hour profiles showed nuanced differences, this specific delay highlights sleep's role in timing incretin secretion.
Normalized GLP-1 concentrations revealed condition-by-time interactions, with post-hoc analyses confirming the breakfast peak shift. This temporal misalignment could underlie sleep loss's metabolic harms, as timely GLP-1 is essential for insulin potentiation and satiety signaling.
Implications for Metabolic Health and GLP-1 Therapies
These results extend evidence that sleep curtailment perturbs incretin pathways, potentially exacerbating obesity and diabetes risks. A delayed GLP-1 peak might prolong hyperglycemia post-meals and heighten hunger, aligning with observed increases in intake after sleep loss.
For patients on GLP-1 agonists, this underscores sleep's synergy with therapy. Poor sleep could blunt endogenous GLP-1 support, reducing overall efficacy. Clinicians might advise 7-9 hours nightly to optimize outcomes in type 2 diabetes or weight loss protocols.
Comparisons to Prior Research
Unlike fragmented sleep studies showing afternoon-only dips, TSD's total deprivation elicited a clearer postprandial delay. Sex differences in restriction studies (females more affected) warrant further male-female comparisons.
Safety Considerations and Patient Guidance
While TSD is extreme, chronic short sleep mimics risks. Patients should discuss sleep with providers, especially if using GLP-1 meds. Tools like Shotlee can help track sleep patterns, symptoms, and medication responses for personalized adjustments. Common side effects of GLP-1 therapies (nausea, GI issues) may worsen with fatigue; monitor closely.
Who might benefit? Those with metabolic concerns—prediabetes, obesity—could prioritize sleep to bolster natural GLP-1. Consult doctors before changes, particularly with comorbidities.
Key Takeaways and Actionable Insights
- Acute total sleep deprivation delays the GLP-1 peak response to breakfast in healthy young men, per Nutrition & Diabetes study.
- 12 participants (age ~22, BMI ~24); controlled 24-hour sessions with standardized meals.
- Breakfast: 600 ml, 112.8 g carbs; GLP-1 assayed via radioimmunoassay.
- Implications: Reinforces sleep's role in incretin function, relevant for diabetes prevention and GLP-1 therapy optimization.
- Practical tip: Aim for consistent 7-9 hours sleep; track with apps for metabolic health.
Conclusion
This study illuminates how even one sleepless night disrupts GLP-1's breakfast response, with broader ramifications for metabolic signaling. By preserving endogenous rhythms, individuals can support GLP-1 pathways naturally, complementing peptide therapies. Future research should explore chronic effects, females, and interventions. For now, prioritize sleep to safeguard incretin health and reduce cardiometabolic risks.